Introduction: Polatuzumab vedotin (Pola) combined with rituximab (R) demonstrated activity and tolerability in a Phase (Ph) II trial of patients (pts) with relapsed/refractory (R/R) diffuse large B-cell lymphoma (DLBCL) (Morschhauser et al. Lancet 2019). The pro-survival MCL-1 protein has been identified as a mechanism of resistance to venetoclax (Ven), a potent inhibitor of BCL-2, in non-Hodgkin lymphoma cell lines. Preclinical studies have demonstrated that concurrent treatment with Pola promotes MCL-1 degradation and enhances anti-tumor efficacy in vivo, thus providing a strong rationale for the combination with Ven (Amin et al. AACR 2020). We sought to determine whether the combination of Pola-Ven-R might further enhance anti-tumor response. Here, we present the primary efficacy and safety analysis from a Ph Ib/II study of Pola-Ven-R in pts with R/R DLBCL (GO29833; NCT02611323).
Methods: GO29833 is an open-label, multicenter study of pts with R/R DLBCL who had received ≥1 prior anti-CD20 chemo-immunotherapy regimen. The recommended Ph II dose (RP2D) combination for Pola-Ven-R was initially defined in a 3+3 dose escalation phase and was then expanded into Ph II. Pts in the expansion cohort received induction therapy with six 21-day cycles of: intravenous (IV) Pola 1.8mg/kg (Cycle [C] 1-6: Day [D]1), Ven 800mg by mouth daily and R 375mg/m2 IV (C1-6: D1). Responders received consolidation therapy for 8 months (Ven 800mg daily and R 375mg/m2 on D1 every 2 months). The primary safety objectives were to determine the RP2D for Pola and Ven when given in combination with R and to evaluate the safety and tolerability of the Pola-Ven-R combination. The primary efficacy endpoint was complete response (CR) at end of induction (EOI), as determined by the Independent Review Committee (IRC) based on positron emission tomography-computed tomography (PET-CT) scans using modified Lugano 2014 response criteria. Secondary objectives included CR-rate at EOI and best overall response (BOR) determined by the investigator (INV). Exploratory objectives included INV-assessed progression-free survival (PFS) and overall survival (OS).
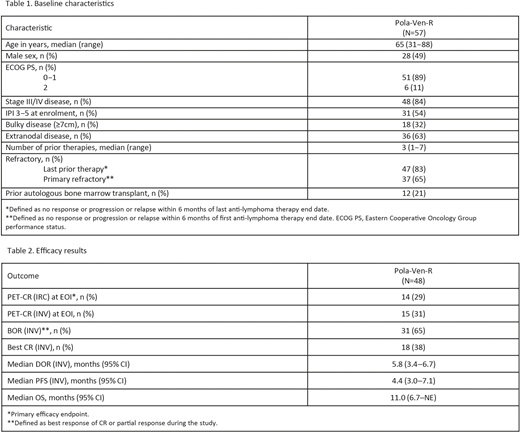
Results: At the primary analysis (January 30, 2020), 57 pts from the Ph Ib/II populations were enrolled and received at least one study drug; the median duration of follow-up was 7.0 (range 0.2-30.4) months. Baseline characteristics of the safety-evaluable pts are shown in Table 1: median age, 65 years; male, 49%; Ann Arbor Stage III-IV, 84%; International Prognostic Index (IPI) ≥3, 54%; median prior lines of therapy, 3; refractory to last line, 83%; primary refractory, 65%. Dose limiting toxicity was not observed in Ph I and the maximum dose level was chosen as RP2D. All except two pts experienced at least one adverse event (AE), 21 (37%) had a serious AE, and 45 (79%) had a Grade 3-4 AE. The most common Grade 3-4 AEs were neutropenia (30 pts, 53%), infections (9 pts, 16%), and anemia (6 pts, 11%). AEs leading to dose reduction or interruption of any drug occurred in 10 (18%) and 35 (61%) pts, respectively; the majority of dose modifications were changes to Ven dosing. Seven (12%) pts had an AE that led to the discontinuation of any study drug (Pola [n=5]; Ven [n=7]; R [n=6]). One grade 5 AE was reported (pneumonia); however, it was not considered related to study treatment as the pt had received new anti-lymphoma therapy following disease progression. In the primary efficacy-evaluable population (n=48), the IRC-assessed modified Lugano CR rate at EOI was 29% (Table 2). The INV-assessed CR rate at EOI and BOR were 31% and 65%, respectively, with a median duration of response of 5.8 months (95% confidence interval [CI]: 3.4-6.7). The median PFS and OS were 4.4 months (95% CI: 3.0-7.1) and 11.0 months (95% CI: 6.7-not evaluable), respectively.
Conclusions: Our study of the novel triplet combination, Pola-Ven-R, demonstrates a safety profile consistent with the known profiles of the individual drugs. This first report of the full efficacy population showed promising activity in a heavily pre-treated and refractory population of pts with R/R DLBCL. Further evaluation of Pola-Ven-R and the impact of consolidation therapy is warranted to address the significant unmet medical need in this patient population.
Gritti:Autolus: Consultancy; Takeda: Honoraria; F. Hoffmann-La Roche Ltd: Honoraria; Amgen: Honoraria; Kite: Consultancy; Italfarmaco: Consultancy; Jannsen: Other: Travel Support; IQVIA: Consultancy. Marlton:Astellas: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; Astra-Zeneca: Membership on an entity's Board of Directors or advisory committees; F. Hoffmann-La Roche Ltd: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; AbbVie: Membership on an entity's Board of Directors or advisory committees. Phillips:Abbvie: Consultancy, Research Funding; Pharmacyclics: Consultancy; Cardinal Health: Consultancy; Bayer: Consultancy, Research Funding; BMS: Consultancy; Seattle Genetics: Consultancy; Incyte: Consultancy, Other: travel expenses; AstraZeneca: Consultancy; Karyopharm: Consultancy; Beigene: Consultancy. Arthur:Royal North Shore Hospital: Current Employment. Bannerji:Regeneron Pharmaceuticals: Research Funding; AbbVie: Research Funding; Sanofi-Pasteur: Other: Spouse is employee; F. Hoffmann-La Roche Ltd/Genentech, Inc and Pharmacyclics LLC, an AbbVie Company: Research Funding. Corradini:Incyte: Consultancy; Celgene: Consultancy, Honoraria, Other: Travel and accommodations paid by for; Janssen: Consultancy, Honoraria; Daiichi Sankyo: Consultancy, Honoraria; Sanofi: Consultancy, Honoraria; KiowaKirin: Consultancy, Honoraria; Servier: Consultancy, Honoraria; BMS: Other; Gilead: Consultancy, Honoraria, Other: Travel and accommodations paid by for; Takeda: Consultancy, Honoraria, Other; AbbVie: Consultancy, Honoraria, Other: Travel and accommodations paid by for; Novartis: Consultancy, Honoraria, Other: Travel and accommodations paid by for; F. Hoffman-La Roche Ltd: Consultancy, Honoraria; Kite: Consultancy, Honoraria; Amgen: Consultancy, Honoraria, Other: Travel and accommodations paid by for. Johnston:MSD: Membership on an entity's Board of Directors or advisory committees; F. Hoffmann-La Roche Ltd: Membership on an entity's Board of Directors or advisory committees. Seymour:Janssen: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; AstraZeneca: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; AbbVie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; F. Hoffmann-La Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Gilead: Consultancy; Mei Pharma: Consultancy, Honoraria; Morphosys: Consultancy, Honoraria; Nurix: Honoraria. Hirata:Genentech, Inc.: Current Employment; F. Hoffmann-La Roche Ltd: Current equity holder in publicly-traded company. Musick:F. Hoffmann-La Roche Ltd: Current equity holder in publicly-traded company; Roche/Genentech, Inc.: Current Employment. Saha:Genentech, Inc.: Consultancy; Genentech, Inc.: Current Employment. Croft:Genentech, Inc.: Current Employment, Current equity holder in publicly-traded company, Divested equity in a private or publicly-traded company in the past 24 months. Flowers:OptumRx: Consultancy; Karyopharm: Consultancy; Gilead: Consultancy, Research Funding; Leukemia and Lymphoma Society: Membership on an entity's Board of Directors or advisory committees; BeiGene: Consultancy; Bayer: Consultancy; Denovo Biopharma: Consultancy; Genentech, Inc./F. Hoffmann-La Roche Ltd: Consultancy, Research Funding; Pharmacyclics/Janssen: Consultancy; Acerta: Research Funding; Spectrum: Consultancy; Millennium/Takeda: Consultancy, Research Funding; TG Therapeutics: Research Funding; Burroughs Wellcome Fund: Research Funding; Eastern Cooperative Oncology Group: Research Funding; National Cancer Institute: Research Funding; AbbVie: Consultancy, Research Funding; V Foundation: Research Funding; Cancer Prevention and Research Institute of Texas: Research Funding; Celgene: Consultancy, Research Funding; Kite: Research Funding.
Polatuzumab vedotin (Polivy) is approved in combination with bendamustine and rituximab or use in third-line or later treatment of relapsed/refractory (R/R) DLBCL in the USA and in second-line or later treatment in the EU. Venetoclax (Venclexta) is approved for the treatment of adult patients with CLL or SLL, and in combination with azacitidine or decitabine or low-dose cytarabine for the treatment of newly-diagnosed AML in adults >75 years, or who have comorbidities that preclude use of intensive induction chemotherapy. Rituximab (Rituxan) is approved for use in relapsed/refractory low-grade NHL and in previously untreated DLBCL with CHOP, but is not approved for use in R/R DLBCL.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal